Josephine Diony Nanda, M.D., Ph.D.
Department of Parasitology, Faculty of Medicine, Public Health, & Nursing (FKKMK), Universitas Gadjah Mada
Cell and gene therapy (CGT) has emerged as one of the most transformative innovations in modern medicine, offering new hope for patients with previously untreatable conditions. In Indonesia, the journey of CGT is still in its early stages, but recent developments highlight a growing commitment to advancing this field. From initial research initiatives and academic workshops to government-led discussions on regulation and infrastructure, Indonesia is beginning to lay the foundation for integrating CGT into its healthcare system. This article explores the recent updates of cell and gene therapy in industry, clinical trials, research, and society (including FARM-related aspects).
Indonesia’s CGT market is expanding rapidly, driven by rising demand for treatments targeting malignancies, genetic disorders, and rare diseases. With strong support from both government initiatives and private investment, the sector is projected to grow steadily through 2031. The Stem Cell & Cancer Institute (SCI) plays a central role in advancing stem cell therapy, cancer research, and diagnostic innovation. Increasing collaboration between pharmaceutical and biotech firms, hospitals, and cancer care centers signals promising progress in CGT development. This momentum is further reinforced by the Ministry of Health and the National Research and Innovation Agency (BRIN), underscoring Indonesia’s commitment to building a robust future for advanced therapies. As these foundations strengthen, the next critical step lies in the initiation and expansion of clinical trials, which will determine how CGT can be safely and effectively integrated into Indonesia’s healthcare system.
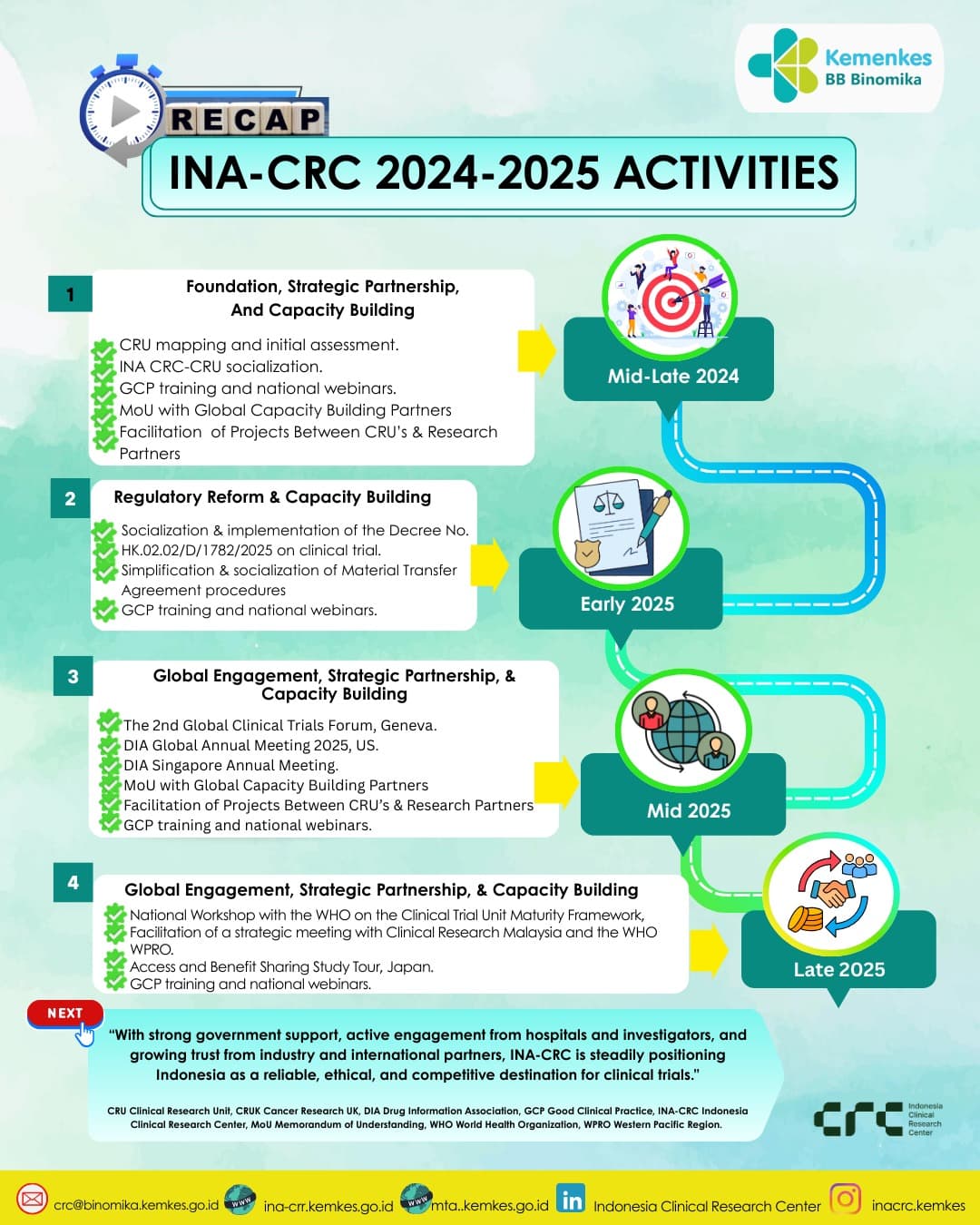
At present, only a small number of active CGT clinical trials are registered in Indonesia for 2024–2025. Most remain in early‑phase (Phase I/II) studies, focusing on stem cell‑based therapies and regenerative medicine. These trials are typically conducted in collaboration between hospitals, universities, and biotech firms, under the oversight of the Ministry of Health and BRIN. To create an environment suitable for further clinical trial development, the Indonesian government established the Indonesia Clinical Research Centre (INA‑CRC) in mid‑2024 to streamline trial processes and attract international sponsors. Since its inception, INA‑CRC has strengthened the regulatory framework for CGT, advanced capacity building, and initiated global partnerships. In late 2025, INA‑CRC, together with the WHO Indonesia Country Office and the WHO Western Pacific Regional Office (WPRO), hosted a national workshop in Jakarta. The workshop focused on strengthening Indonesia’s clinical trial ecosystem and piloting the WHO Clinical Trial Unit‑Maturity Framework (CTU‑MF), aligning with the Global Action Plan for Clinical Trial Ecosystem Strengthening. The establishment of INA‑CRC and Indonesia’s move to WPRO in 2025 mark a major step forward in clinical trial readiness, creating stronger opportunities for cell and gene therapy trials as Indonesia aligns with international standards and expands regional collaboration. Building on this foundation, Indonesia is now entering the next phase: advancing CGT research to translate these early efforts into scientific breakthroughs and clinical applications.
Research in Indonesian cell and gene therapy (CGT) has largely concentrated on the application of stem cells in oncology, cardiovascular conditions, degenerative disorders, and infectious disease studies. To foster innovation, the National Research and Innovation Agency (BRIN) partnered with the Indonesian Stem Cell Association (ASPI) to organize international seminars promoting advances in stem cell research and regenerative medicine. One such event, titled Future Directions and Opportunities in Stem Cell Innovation for Clinical Application and the Health Industry, took place at Gedung B.J. Habibie BRIN in Jakarta on August 5–6, 2025. This collaboration was intended to accelerate the transition of research from the laboratory to industrial application. The seminars were followed by a continuous technical workshop designed to enhance researchers’ capacity in bridging scientific findings with healthcare practice.
In general, while sentiments toward new CGT innovation are largely positive, some concerns remain regarding their long-term safety. This underscores the need for continued research and clinical trials to validate both their benefit and lasting impact. Innovation in CGT is not limited to healthcare; gene-editing technologies are also being explored in food and agriculture to enhance food security and crop resilience. Yet, public debate persists: scientists emphasize opportunities for sustainable farming and improved nutrition, whereas skeptics raise concerns about biodiversity and the potential health implications of GMO crops or lab-produced food.
Although CGT still has a long journey before becoming widely established and accessible, it remains a promising pursuit. With the support of the Indonesian government, various research institutes, universities, and industries are working collaboratively to advance CGT to the future where CGT can be utilized fully to enhance quality of life of Indonesian citizens.

Leave a comment