Kitipong Uaesoontrachoon1, Parin Rattananon1, Usanarat Anurathapan1,2, Suradej Hongeng1
1Genepeutic Bio, Co. Ltd., Bangkok, Thailand
2Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand
Abstract
Gene therapy has emerged as a transformative therapeutic modality for genetic and hematologic diseases. In Thailand, where hemoglobinopathies and pediatric acute lymphoblastic leukemia (ALL) represent significant public health burdens, the development of domestic gene therapy capacity presents both clinical opportunity and economic challenge. This manuscript examines Thailand’s healthcare financing structure, disease burden, research ecosystem, regulatory preparedness, and emerging industrial capacity in gene and cell therapies. While hematopoietic stem cell transplantation (HSCT) remains the only curative option for severe beta-thalassemia and relapsed ALL, gene therapy offers the potential for durable remission and long-term cost offsets. However, uncertainties regarding long-term efficacy, affordability, regulatory readiness, and workforce preparedness remain substantial. We discuss Thailand’s strategic investments under the Thailand 4.0 framework, the establishment of local GMP-certified manufacturing infrastructure, and the regulatory evolution surrounding Advanced Therapy Medicinal Products (ATMPs). The analysis highlights both the promise and systemic challenges of positioning Thailand as a regional gene therapy hub within ASEAN.
Key words: Advanced Therapy Medicinal Products, CAR-T cell Therapy, Refractory/relapsed ALL, Adoptive Cell Immunotherapy
Introduction
Gene therapy has experienced renewed global momentum, driven by advances in vector design, genome editing technologies, and chimeric antigen receptor T-cell (CAR-T) therapies. Genetic diseases arise from defective genes that result in dysfunctional or absent protein production. Therapeutic correction at the genetic level represents a potentially curative approach rather than symptomatic management. To date, there have been 32 gene therapies approved worldwide by the US FDA (US FDA website), with more than 2,000 therapies in development, ranging from preclinical stages to pre-registration (American Society of Gene and Cell Therapy, 2023). Based on the current landscape, gene therapies are anticipated to become a key component of mainstream medical care in the near future, though restricting in the high-income countries due to the high initial treatment price.
For middle-income countries such as Thailand, the burden of genetic disorders extends beyond clinical morbidity. High-prevalence hereditary diseases impose sustained financial pressures on public health systems and reduce long-term economic productivity. Consequently, effective gene therapy—if made accessible and cost-effective—could significantly alleviate both healthcare expenditure and societal burden.
Disease Burden in Thailand
Beta-Thalassemia and Hemoglobinopathies
Hemoglobinopathies are the most prevalent inherited disorders in Southeast Asia. In Thailand, approximately 3,000 infants are born annually with severe beta-thalassemia (Fucharoen et al, 2012). Patients require lifelong monthly blood transfusions to survive. Without transfusion support, survival is limited to only a few years.
Chronic transfusion therapy is associated with several challenges:
- Blood shortages and screening costs
- Iron overload leading to organ failure
- Requirement for intensive iron chelation therapy (8–12 hours daily, 5–7 days per week)
These treatment burdens significantly reduce quality of life and generate sustained healthcare costs (Riewpaiboon et al, 2010).
Acute Lymphoblastic Leukemia (ALL)
Acute lymphoblastic leukemia (ALL) is the most common pediatric malignancy worldwide and in Thailand. Between 2006 and 2011, Thailand reported an age-standardized incidence rate of 38 per million children, of which 28 per million were ALL cases (Tharnprisan et al, 2013). B-cell ALL accounted for 83.5% of cases. Peak incidence occurs between ages 2–5 years, representing over 75% of cases (Wiangnon et al, 2014).
Currently, hematopoietic stem cell transplantation (HSCT) is the only established curative therapy for both severe beta-thalassemia and relapsed ALL.
Health Economics and Public Financing
Thailand’s Universal Coverage Scheme (UCS) provides comprehensive health coverage for its citizens which has become a key element in the national health infrastructure. The UCS provides medical access to approximately 76% of Thailand’s population. The scheme offers free healthcare services, encompassing outpatient, inpatient, and emergency care, to uninsured individuals not covered by the two other main health insurance programs: the Civil Servant Medical Benefit for government employees and retirees and the Social Health Insurance for private sector employees (Tangcharoensathien et al, 2018). These established prevention and treatment strategies are efficiently administered and remain affordable within the country’s fiscal spaces.More than 600,000 thalassemia patients receive government-funded care at an estimated cost of approximately US$3,000 per patient per year (NHSO, 2021).
For patients undergoing HSCT, the government provides approximately US$21,000 to cover treatment and monitoring during the first year surrounding transplantation (Leelahavarong et al, 2010). Cost-effectiveness analyses suggest that HSCT is economically justified for patients younger than 10 years of age.
However, gene therapies are typically associated with high upfront costs. The excessively high cost of gene therapies could undermine the willingness of government funders to cover the expenses, potentially reducing the availability of services for other patients within the healthcare programs. The individual price tag of $2.8 million for betibeglogene autotemcel is nearly identical to the entire $2.91 million fiscal year budget utilized by the UCS in 2021 to provide hematopoietic stem cell transplantation (HSCTs) to 107 individuals with leukemia, lymphoma, and thalassemia (NHSO, 2023). Although they may provide long-term savings by reducing lifetime treatment expenses, uncertainty regarding durability of response complicates reimbursement decisions. Therefore, experts advocate for coverage with evidence development (CED), whereby reimbursement is conditional upon long-term outcome data collection (Mohara et al, 2012).
Such models require:
- Coordination among multiple health insurers
- National registries and data-sharing mechanisms
- Transparent governance of long-term clinical data
Potential conflicts may arise if proprietary registries are controlled by private gene therapy companies.
National Research and Innovation Strategy
Thailand launched its “Thailand 4.0” strategy in 2017, aiming to increase R&D expenditure to 4% of GDP by 2036. As of 2018, R&D spending stood at 1.11% of GDP—below the averages for upper-middle-income (1.41%) and high-income countries (2.43%; NXPO, 2019).
To strengthen competitiveness, Thailand established (Yuthavong et al, 2019):
- Thailand Towards Excellence in Clinical Trials (ThaiTECT)
- Incentives for private-sector R&D investment (up to 15-year tax exemptions; BOI, 2023)
- Technology and Innovation Implementation Programs supporting SMEs
- Talent Mobility programs linking academia and industry
Despite these advances, continuing professional education in advanced health biotechnology remains limited. Strengthening domestic expertise and incentivizing overseas-trained Thai professionals to return may be critical to sustaining gene therapy innovation.
Clinical Development and Industrial Capacity
Thailand has begun participating in international gene therapy clinical trials for beta-thalassemia in collaboration with a US company called Bluebird Bio. In parallel, domestic infrastructure development has accelerated.

Figure 1. Thailand’s First GMP-Certified ATMP Manufacturing Suite. Located at Genepeutic Bio, this facility achieved the historic first Thai FDA certification for CAR T-cell manufacturing in 2023. The infrastructure is designed specifically to support the production of Advanced Therapy Medicinal Products (ATMPs) under international PIC/S GMP standards, providing the industrial backbone for the country’s “Medical Hub” initiative.
Genepeutic Bio Co., Ltd, established in 2020, represents Thailand’s first GMP-certified gene therapy manufacturing facility (Figure 1). The organization focuses on bridging the gap between advanced biomedical research and clinical accessibility for patients with hard-to-treat cancers.With partial governmental support, GNPT aims to locally produce CAR-T cell therapies and gene therapies.
Clinical Focus: The lead pipeline programs target CD19-positive B-cell malignancies, including Acute Lymphoblastic Leukemia (ALL) and Diffuse Large B-cell Lymphoma (DLBCL). Its CAR-T construct has secured both proprietary interllecture property (IP) and the necessary freedom to operate (FTO). This ensures that clinical translation and commercial scaling are not hindered by international patent litigation—a common barrier for biotech firms in middle-income countries.
Manufacturing Excellence: By utilizing automated closed-system processing, Genepeutic Bio minimizes contamination risks and ensures the scalability of highly complex cell products.
- While currently approved CD19 CAR-T cell products globally are primarily restricted to the autologous setting—relying on the genetic modification of a patient’s own T-cells—this approach presents substantial logistical and clinical challenges. Autologous manufacturing is often hindered by the poor fitness of T-cells from heavily pre-treated patients, as well as the high cost of individualized, small-batch production.
- To address these limitations, Genepeutic Bio is developing a pioneering allogeneic platform. This strategy utilizes T-cells from healthy donors rather than the patients themselves, offering several systemic advantages:
- Donor Selection and HLA-Matching: The methodology utilizes either HLA-matched or haploidentical (half-matched) donors as the source of the starting cellular material. This allows for rigorous pre-screening and selection of high-potency immune cells.
- Scalability and Accessibility: Allogeneic products can be manufactured in large batches, cryopreserved, and shipped on demand. This eliminates the 3–4 week manufacturing wait time inherent in autologous processes, which is critical for patients with rapidly progressing malignancies.
- Historic Milestone: If approved by the Thai FDA, this would represent the first clinical product to successfully utilize an allogeneic method for CAR-T cell treatment in the region, positioning Thailand at the global forefront of cellular immunotherapy innovation.
The establishment of Genepeutic Bio represents a shift in the regional healthcare landscape:
- Local Self-Reliance: Reducing dependence on expensive, logistically complex imports of cell and gene therapies from the West.
- Cost Accessibility: Drastically lowering the “price-per-dose” to make life-saving treatments viable for the ASEAN patient population.
- Regulatory Leadership: Collaborating closely with the Thai FDA to establish the gold standard for Advanced Therapy Medicinal Products (ATMPs) in the region by adopting the international standards.
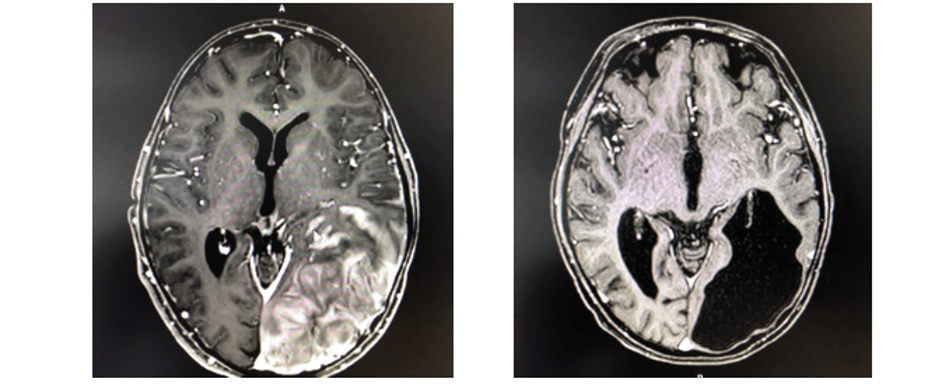
Figure 2. Magnetic Resonance Imaging (MRI) demonstrating regression of leukemic brain infiltration after haploidentical CD19 CAR-T cells infusion resulting in remission of 18 months.
Key Milestone: In 2023, Genepeutic Bio successfully received the first-ever Thai FDA certification for a CAR T-cell manufacturing site, marking a historic leap for Thailand’s “Medical Hub” initiatives and initiated its first-in human clinical trial.
In a landmark proof-of-concept evaluation, domestic CAR-T therapy was successfully administered to an 11-year-old patient facing an exceptionally high-risk clinical scenario. The patient presented with B-cell ALL in its fourth relapse, complicated by extramedullary central nervous system (CNS) involvement—a condition traditionally associated with poor prognosis and limited therapeutic options. This complexity was further compounded by a persistent minimal residual disease-positive (MRD+) status and a history of prior Haploidentical Stem Cell Transplantation.
Notably, the haploidentical allogeneic CAR-T cell infusion demonstrated potent therapeutic efficacy by successfully clearing the leukemic cells within the CNS (Figure 2). The treatment was well tolerated, achieving MRD-negative complete remission within 30 days of administration. Longitudinal data from this initial cohort using allogenenic setting reflects the robust durability of the response, with overall survival (OS) and leukemia-free survival (LFS) rates of 70% and 80%, respectively (Figure 3).
Clinical trial evaluating mainly the safety profile (Phase I) of the company’s first product and the very first in human study for gene therapy in Thailand was completed in 2025. The safety profile and dose escalation were demonstrated a manageable safety profile across dose levels evaluated in the dose-escalation phase (Thai Clinical Trials Registry: TCTR20220624004; unpublished data).
- Dose-Limiting Toxicities (DLTs): No DLTs were observed within the predefined 28-day evaluation window following infusion at either dose level.
- Adverse Events of Special Interest: While incidents of Cytokine Release Syndrome (CRS) and Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) were monitored according to ASTCT (American Society for Transplantation and Cellular Therapy) consensus grading, all events were clinically manageable with standard institutional protocols (e.g., tocilizumab and/or corticosteroids).
- Tolerability: The current safety data supports the continued clinical development and potential dose expansion of GNPTCD19SPA-0001 in this highly pre-treated patient population.
- Secondary outcome analysis was conducted on an initial cohort ofevaluable patients with relapsed/refractory B-cell malignancies. Preliminary data indicates a potent therapeutic signal with
- Objective Response Rate (ORR): 100%.
- Complete Remission with Incomplete Hematologic Recovery (CRi): 100%.
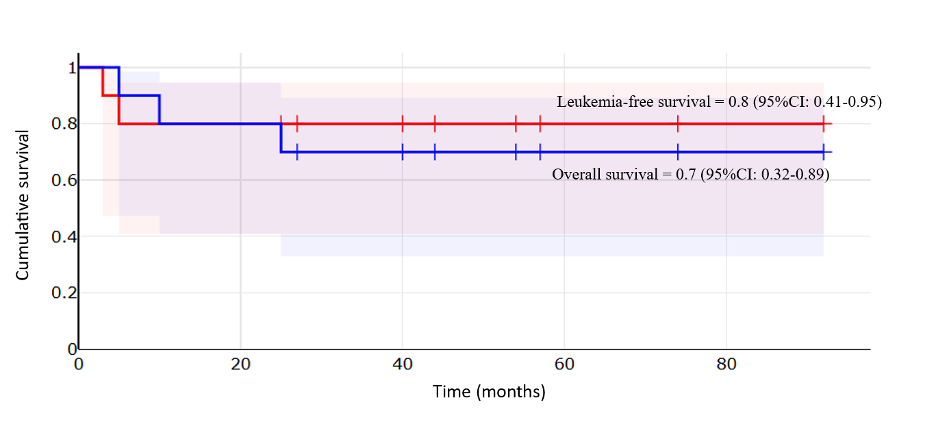
Figure 3. Kaplan-Meier analysis of Overall Survival (OS; 70%) and Leukemia-Free Survival (LFS; 80%) in patients with relapsed/refractory B-ALL treated allogeneic CD19 CAR-T cells.
These high rates of molecular and hematologic response, even at the lower dose cohorts, suggest that the locally manufactured GNPTCD19SPA-0001 construct possesses robust in vivo expansion and anti-tumor effector function. The clinical development of Thailand’s first domestic CAR-T therapy has reached a critical juncture. In March 2026, the company officially initiated its Phase II clinical trial, building on the high efficacy and safety profile established in earlier proof-of-concept evaluations.
Under the current regulatory framework, which emphasizes “Fast-Track” access for rare life-saving therapeutics, the trial is strategically positioned for a rapid conclusion. The company expects to complete this phase and secure conditional approval from the Thai FDA by the end of 2026. This milestone would mark a historic shift in the Thai healthcare landscape, transitioning from experimental research to the official provision of locally manufactured, high-value cellular immunotherapy.
Regulatory Framework and Policy Evolution
In 2018, Thailand introduced regulations governing Advanced Therapy Medicinal Products (ATMPs), including gene and cell therapies. However, regulatory capacity remains under development, particularly regarding:
- Manufacturing oversight
- Product traceability
- Quality assurance standards
- Long-term pharmacovigilance
Thailand aims to harmonize its regulatory framework with established systems in the United States, European Union, Japan, and Australia. Risk-based and accelerated approval pathways may be appropriate for life-threatening diseases lacking alternative treatments (Yuthavong et al, 2021).
Close collaboration between regulators and pioneering domestic companies will be essential to building regulatory expertise in personalized medicine.
Regional Positioning and Medical Tourism
Thailand has historically positioned itself as a regional leader in medical tourism. With regulatory maturation and manufacturing capability, gene therapy could become a high-value extension of this sector.
Strategic Shift to High-Value Healthcare
Under the Thailand 4.0 framework and the Strategic Plan for Thailand: A Hub of Wellness and Medical Services (2016–2025/2026), the Thai government is shifting its medical tourism focus from elective cosmetic and dental procedures to “High-Value Healthcare” such as Advanced Therapy Medicinal Products (ATMPs). The inclusion of gene and cell therapies is projected to generate an additional 1.5 billion THB (approx. US$42 million) in annual revenue by 2026 (MOPH, 2025).
- Regional Dominance: Thailand currently leads ASEAN in medical tourism, but the domestic production of CAR T-cells by Genepeutic Bio provides a unique competitive edge against regional peers (e.g., Singapore, Malaysia) by offering world-class therapy at a significantly lower “price-per-dose.”
- The “Medical Valley” Initiative: The development of specialized zones, such as the EECmd (Eastern Economic Corridor of Medicine) and the Phuket International Medical Tourism Complex (Medical Plaza), aims to integrate advanced cell therapy clinics with luxury recovery services for international patients.
Genepeutic Bio as a Gateway for International Patients
As the first GMP-certified for cell and gene therapy company in Thailand, Genepeutic Bio serves as the industrial backbone for this “Medical Hub” policy:
- Cost Competitiveness: While CAR T-cell therapies in the United States or Europe can cost upwards of US$375,000–$475,000, domestic manufacturing in Thailand aims to provide these treatments at a fraction of the cost, attracting “self-pay” patients from across the Asia-Pacific and the Middle East.
- Inbound Partnerships: The company is actively fostering international corridors to streamline patient referrals and technology transfers between Thailand and overseas.
- Visa Facilitation: To support these high-complexity cases, the Thai government has introduced specialized Medical Visas that allow for extended stays (up to 90 days, extendable) for patients undergoing long-term monitoring post-infusion.
The integration of domestic ATMP manufacturing into Thailand’s ‘Medical Hub’ policy marks a strategic evolution toward high-value medical tourism. By leveraging the lower cost-base of Genepeutic Bio’s CAR T-cell production, Thailand is positioned to capture a significant share of the regional oncology market, specifically targeting patients from the GCC and Asia-Pacific who seek affordable, international-standard gene therapies (Bangkok Post, 2025; ArokaGO, 2025).
Policy Implications and the Path Forward
Thailand’s experience highlights broader lessons for LMICs:
- High upfront cost is the central barrier to gene therapy access.
- Local manufacturing and technology transfer may significantly reduce prices.
- Regulatory readiness must evolve alongside industrial capacity.
- Strategic purchasing alone cannot absorb multimillion-dollar therapies without structural innovation.
- Regional ASEAN collaboration could distribute infrastructure costs and expand patient pools.
By leveraging universal health coverage, prior success in integrating expensive treatments, and growing biotechnology capacity, Thailand could emerge as a regional gene therapy hub.
Conclusions
Gene therapy presents a transformative opportunity for Thailand to reduce the long-term burden of hemoglobinopathies and pediatric leukemia while strengthening its biomedical innovation ecosystem. Strategic investments in regulation, reimbursement models, workforce development, and local manufacturing will determine whether Thailand can successfully transition from participant to regional leader in advanced therapeutics.
Thailand stands at a pivotal moment in the development of domestic gene therapy capacity. The country benefits from:
- Universal health coverage
- Strong government commitment to innovation
- Emerging GMP-certified manufacturing infrastructure
- Growing clinical research capacity
However, major challenges persist:
- High upfront costs and uncertain long-term efficacy
- Regulatory readiness and pharmacovigilance capacity
- Workforce training gaps
- Sustainable reimbursement mechanisms
If addressed systematically, Thailand could emerge as a regional leader in gene therapy development and delivery within ASEAN.
Corresponding author:
Kitipong Uaesoontrachoon, PhD
Genepeutic Bio, Co. Ltd
2Fl., INC2 Building, Tower C, 141 Thailand Science Park, Phaholyothin Road, Klong 1, Klong Luang, Patumthani, 12120 Thailand
Tel +66-632655398
e-mail: Kitipong.uae@gnptbio.com

Dr. Kitipong Uaesoontrachoon is a leading scientist in genetic medicine and currently serves as the Chief Scientific Officer of Genepeutic Bio, Thailand’s first GMP-certified cell and gene therapy manufacturing facility. He earned his PhD in Molecular and Cellular Biology from the University of Melbourne and completed postdoctoral training at the Children’s National Medical Center. Specializing in neuromuscular diseases, particularly Duchenne Muscular Dystrophy, he has authored more than 50 peer-reviewed publications and contributed significantly to therapeutic development in this field.
Prior to his current role, Dr. Uaesoontrachoon served as Principal Director of Research at AGADA Biosciences in Canada and held an adjunct professorship at Dalhousie University. He is also a key figure in Thailand’s national “Medical Hub” initiative, leading Genepeutic Bio to obtain the first certification from the Thai Food and Drug Administration for a CAR-T cell manufacturing facility and to initiate first-in-human clinical trials for pediatric acute lymphoblastic leukemia (ALL). Through this work, he has played an important role in advancing Thailand’s capabilities in advanced therapy medicinal products (ATMPs) and strengthening the bridge between global research standards and local clinical accessibility.
Disclosure: The author declares that there is no relevant or material financial interests that relate to the information described in this paper. Genepeutic Bio is a private clinical stage biotech company for the manufacturing of cell and gene therapy product (CAR-T cells) for oncologic diseases. The company also receives funding from Program Management Unit for Competitiveness (PMUC) for its clinical stage development of CAR-CD19 T-cells for Acute Lymphocytic Leukemia (ALL).
References
- American Society of Gene and Cell Therapy. Gene, cell, an RNA therapy landscape: Q1 2023 quarterly report; 2023. Avaliable from: https://asgct.org/global/documents/asgct-citeline-q1-2023-report.aspx.
- Wiangnon S, Hongeng S, Freechaoon S, Chotsampancharoen T, Jinamoy S. Childhood cancer incidence and survival in Thailand: a report from the Thai Pediatric Oncology Group (ThaiPOG). Jpn J Clin Oncol. 2014 Dec;44(12):1158-67.
- Tangcharoensathien V, Witthayapipopsakul W, Panichkriangkrai W, Patcharanarumol W, Mills A. Health systems development in Thailand: a solid platform for successful implementation of universal health coverage. Lancet. 2018;391(10126):1205-23. Epub 2018/02/06. doi: 10.1016/s0140-6736(18)30198-3. PubMed PMID: 29397200.
- Fucharoen S, Weatherall DJ. The hemoglobinopathies: the Southeast Asian experience. Int J Hematol. 2012 Jun;95(6):594-600.
- Riewpaiboon A, et al. Cost of care for thalassemia patients in Thailand. Pharmacoeconomics. 2010 Oct;28(10):907-19.
- Tharnprisan P, Khiewyoo J, Sripraya P, Wiangnon S. Relapse-free rate with childhood acute lymphoblastic leukemia treated under the thai national protocol. Asian Pac J Cancer Prev. 2013;14(2):1127-30.
- National Health Security Office (NHSO). NHSO Annual Report Year 2021. Bangkok: NHSO; 2022 Oct 21 [cited 2023 Jun 24]. Available from: https://eng.nhso.go.th/view/1/Annual_Reports/EN-US.
- National Health Security Office (NHSO). NHSO Annual Report Year 2021. 2022 October 21[cited 2023 Jun 24]; Available from: https://eng.nhso.go.th/view/1/Annual_Reports/EN-US.
- Leelahavarong P, Teerawattananon Y, Yenruedee P. A cost-utility and budget impact analysis of allogeneic hematopoietic stem cell transplantation for severe thalassemic patients in Thailand. BMC Health Serv Res. 2010;10:209.
- Mohara A, Riewpaiboon A, Teerawattananon Y. Advanced health biotechnologies in Thailand: redefining policy directions. J Med Ethics Hist Med. 2012;5:2.
- Office of National Higher Education Science Research and Innovation Policy Council (NXPO). Thailand’s R&D Expenditure Survey Report 2018. Bangkok: Ministry of Higher Education, Science, Research and Innovation; 2019.
- Yuthavong K. Present clinical trial regulatory system and strategies for improvement in Thailand. Thai J Pharm Sci. 2021;45(2):121-8.
- Board of Investment (BOI). A Guide to the Board of Investment 2023: Incentives for Research and Development. Bangkok: Office of the Prime Minister; 2023.
- Ministry of Public Health (MOPH) Thailand. Thailand ATMP Roadmap 2025–2029: Fast-Track to Access & Innovation. Strategic Dashboard. Nonthaburi: MOPH; 2025 [cited 2026 Mar 4]. Available from: ops.moph.go.th.
- ArokaGO. Public-private partnership supports ATMPs to build a billion-dollar health economy. 2025.
- Bangkok Post. All eyes on Thailand! Advancing ATMPs-Stem Cell toward high-level medicine. Bangkok Post. 2025.
Leave a comment