Joseph Wangisani Chisaka
Department of Biochemistry and Molecular Cell Biology, Taipei Medical University, Taipei, Taiwan
Introduction
Extracellular vesicles (EVs) are emerging as powerful tools for targeted therapy of a wide range of pathologies through functional transfer of biomolecular cargoes, such as nucleic acids, lipids and proteins, to recipient cells and regulate their downstream signaling pathways to modulate their fate. As naturally occurring delivery vehicles, EVs possess a number of advantageous characteristics, including unique size and structure, excellent biocompatibility, immunologically inert, increased stability in circulation, intrinsic targeting capacity and the capability of membrane fusion and crossing of biological barriers (Cai et al., 2024)(Lu et al., 2023). According to inquiries in the NCBI PubMed database, there has been an increase of 211% in EVs publication between the 5 years period of 2019–2024 (8,145 publications) compared to the 5 years period between 2014–2019 (3,113 publications), with a relatively higher fraction of the publications investing the transport of small molecules, macromolecular complexes, and nucleic acids (Figure 1). This reflects a booming research interest and EVs widening application potential in therapeutics and diagnostics (Li et al., 2025).
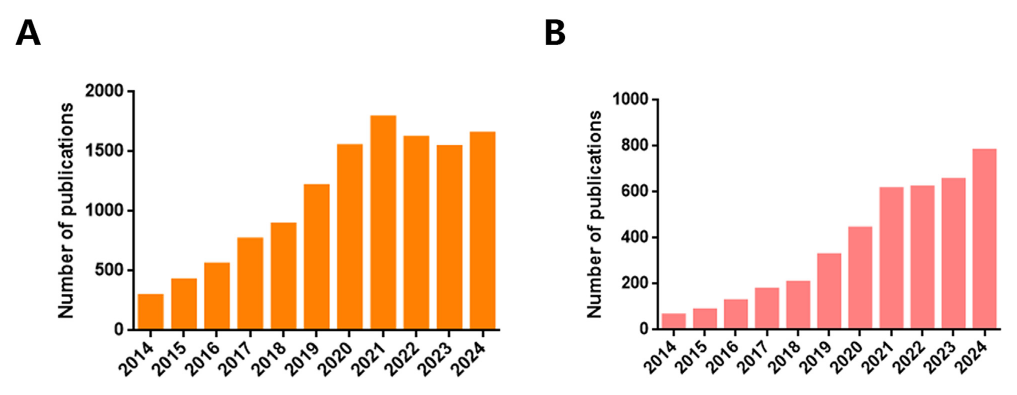
Figure 1: The number of Extracellular vesicles publications between 2014 and 2024 searched by (A) “biomarkers” and (B) “drug delivery” keyword, according to PubMed.
Copied from: Li Ziqian et al., (2025); DOI 10.3389/fbioe.2025.1487627
However, despite the great potential for their clinical application, challenges with cargo loading and EVs low yield continue to be reported as significant obstacles for EVs transitioning from experimental research to clinical practice. As such, strategies for enhancing EV cargo-loading and promoting EV release would have broader application prospects with substantial outcomes. This has resulted in recent growing interest in engineering EVs to specifically alter their cargo. Although concerns have been raised regarding the natural body processes which function to eliminate foreign protein or nucleic acid drugs whenever they are introduced into the body by the immune response. However, in the case of the EVs, the vesicle structure acts as a protection cover for the drug molecules against internal immune elimination (Wang et al., 2025).
EVs engineering
The delivery properties of EVs can be improved by genetic engineering of parent cells (endogenous modification) or direct modification of the EVs (exogenous modification). In the endogenous modification, parent cells are altered before EVs are isolated to make the secreted EVs carry specific molecules. For example, a vector containing the target gene can be transfected into parent cells using genetic engineering technology. The parent cells then produce and secrete proteins or ncRNAs encoded by the inserted genes and package them into EVs through a natural packaging process (Lu et al., 2023)(Wang et al., 2025). EVs enriched with target proteins or nucleic acids can therefore be obtained by purifying the cell culture supernatant. This EV biosynthesis system is limited based on the fact that the sorting system utilized by the cells to determine which substances are packaged into EVs is so complex. As a result, this limits specificity and often result in random packaging into EVs and low loading efficiency (Wang et al., 2025).
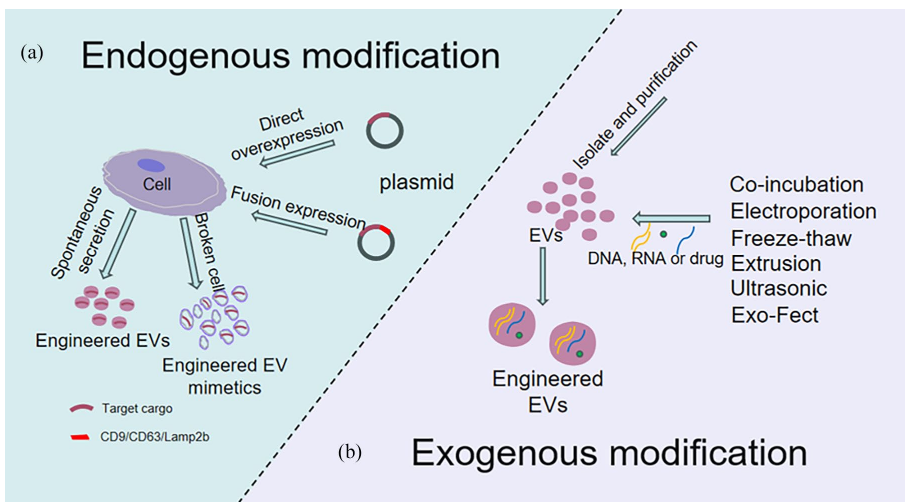
Figure 2: Endogenous and exogenous extracellular vesicle modification.
Copied from: Wang Yuxuan et al (2025); Journal of Tissue Engineering. DOI: 10.1177/20417314251319474
On the other hand, exogenous modification involves loading target molecules directly onto isolated and purified EVs through different engineering techniques which include co-incubation, electroporation, extrusion, ultrasonic treatment, calcium chloride-mediated transfection, saponin permeabilization, freeze-thaw cycles etc. (Wang et al., 2025).
Recent advances in functional EV engineering
As organic nanoparticles, functional engineered EVs generally exhibit lower toxicity and fewer adverse reactions. However, they often suffer from poor stability and suboptimal biodistribution, requiring many modifications to achieve effective cargo delivery. Below are some of the engineering techniques adopted in recent researches to address some of the EVs engineering challenges earlier discussed:
Microfluidics: A promising technology which enables the use of small devices and minimal sample amounts for exosome collection, loading and detection while providing ease of scalability compared to traditional methods. Researchers have recently employed this technology to facilitate the generation, modification, and loading of exosomes and exosome-mimetic nanovesicles in a highly efficient and precise manner.
Nanopration: Another innovative technology that employs nanofluidic devices with nanochannel electroporation and enables efficient loading of specific cargos into exosomes with high effectiveness. This technology has already shown success in various preclinical models despite being recently developed and has offered relatively similar advantages to those of microfluidic systems.
Genetic engineered sorting: Cargo loading is achieved prior to exosome isolation under this technology, which minimizes loss or damage to exosomes. It preserves the structural integrity of exosomes from exposure to freeze–thaw cycles and destabilizing temperatures as is the case with electroporation techniques. However, due to the genetic engineering constraints, this method is only suitable for cargos naturally produced within cells, thus excluding small molecule drugs (Liu et al., 2025).
Engineered EVs in clinical trials
Advances in EVs engineering have enabled precise therapeutic delivery and investigation in clinical trials. A survey on Clinical Trials.gov (https://clinicaltrials.gov/) shows the major applications of exosomes are biomarkers, exosome-therapy, drug delivery systems, and cancer vaccines. An analysis shows that a total of 116 trials have been recorded of which 6 (5.17%) studies were for drug-delivery system trials (Rezaie, Feghhi and Etemadi, 2022). An on-going phase I trial is investigating the intravenous delivery of siRNA-loaded Bone Marrow MSC-derived exosomes targeting KRAS G12D mutations in metastatic Pancreatic Ductal Adenocarcinoma (PDAC) patients who failed multiple lines of therapy (NCT03608631). Another on-going phase II trial is investigating whether sub-endometrial injection of mechanically engineered umbilical cord-derived stem cell exosomes can improve endometrial thickness or clinical pregnancy rates compared to platelet-rich plasma in severe intrauterine adhesions following adhesiolysis (NCT06896747).
Conclusion
EVs have generated massive interest as a promising therapeutic option on a wide range of diseases with unmet needs through their demonstrated high biocompatibility and low immunogenicity as natural cell products, stability due to the lipid bilayer that protects from enzymatic degradation, and the ability to cross biological barriers which enhance their targeting capabilities. On the other hand, engineered EVs can further provide enhanced precision in targeted disease therapeutics. While EVs may be synthesized in Good Manufacturing Practice (GMP) settings, significant issues related to immunogenicity (especially engineered EVs), safety, toxicity, biodistribution, and targeting persist.
References
Cai, H. et al. (2024) ‘Delivering synaptic protein mRNAs via extracellular vesicles ameliorates cognitive impairment in a mouse model of Alzheimer’s disease’, BMC Medicine, 22(1), pp. 1–18. doi:10.1186/s12916-024-03359-2.
Li, Z. et al. (2025) ‘Advancements in extracellular vesicles biomanufacturing: a comprehensive overview of large-scale production and clinical research’, Frontiers in Bioengineering and Biotechnology, 13(February), pp. 1–19. doi:10.3389/fbioe.2025.1487627.
Liu, J.J.J. et al. (2025) ‘Exosomes in cancer nanomedicine: biotechnological advancements and innovations’, Molecular Cancer, 24(1). doi:10.1186/s12943-025-02372-0.
Lu, M. et al. (2023) ‘Extracellular vesicle-based nucleic acid delivery’, Interdisciplinary Medicine, 1(2). doi:https://doi.org/10.1002/INMD.20220007.
Rezaie, J., Feghhi, M. and Etemadi, T. (2022) ‘A review on exosomes application in clinical trials: perspective, questions, and challenges’, Cell Communication and Signaling, 20(1), pp. 1–13. doi:10.1186/s12964-022-00959-4.
Wang, Y. et al. (2025) ‘Extracellular vesicles : From large-scale production and engineering to clinical applications’. doi:10.1177/20417314251319474.
Leave a comment