Jui-Chien Hsing
Taipei Medical University, Taipei
The 2025 APSEV International Conference, held in Singapore, spotlighted major breakthroughs at the frontier of regenerative nanomedicine. As a leading event in Asia devoted to extracellular vesicle (EV) research, APSEV provided key insights into how these vesicles—particularly engineered cell-derived nanovesicles (CDNs)—are transitioning from being seen as passive cellular debris to becoming advanced therapeutic tools. With growing relevance in tissue repair, oncology, and regenerative medicine, EVs are now redefining precision therapy in modern medicine.



The era of engineered CDNs has arrived
A significant aspect of the presentation was the emergence of engineered cell-derived nanovesicles (CDNs) as versatile carriers capable of delivering RNA, proteins, and therapeutic agents with high specificity. Rather than remaining purely theoretical, methodologies such as membrane remodeling, modulation of vesicle permeability, and optimization of miRNA payloads have been effectively translated into reproducible experimental protocols. Notably, CDNs loaded with specific microRNAs exhibited considerable cytoprotective effects, particularly in reducing oxidative stress and preventing cell death in injury models. These results highlight a pivotal shift within the field, transforming extracellular vesicles (EVs) from passive biological byproducts into purposefully designed, bioactive delivery systems.
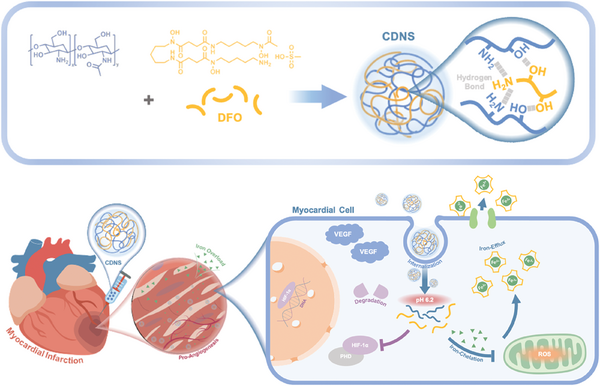
CDNs structure diagram [1]
From “Delivery Vehicle” to “Targeted Navigator”
What truly marked a leap forward was the integration of peptide-guided delivery strategies along with click chemistry. One prominent study showed cDNA functionalized using IMTP or Ischemic Myocardium-Targeting Peptide. These bioconjugation reactions were used for performing the functionalization without copper. Researchers used Ac4ManNAz-based metabolic labeling; in addition, they then used SPAAC (strain-promoted azide-alkyne cycloaddition); through this, researchers precisely presented IMTP on cDNA plus did not compromise vesicle integrity or function. This bioengineering approach enabled cDNA for homing in on infarcted heart tissue because it demonstrated improved biodistribution as well as therapeutic localization in vivo. Such targeted delivery minimizes off-target effects plus unlocks a new modular platform applicable for disease-specific uses, setting the stage for next-generation precision nanomedicine.
A Reflective Parallel—miR-24-3p in the Ovary and Heart
Among all of the sessions, one presentation showed that miR-24-3p protects the heart in a deep way for me. The mechanism is similar to my own findings within a chemotherapy-induced POI model. MiR-24-3p suppresses Keap1 to activate the Nrf2/ARE antioxidant pathway, also this likewise reduces oxidative damage and cell death in ovarian tissue. We may be able to achieve localized non-hormonal regenerative treatment of POI and related reproductive disorders if cDNAs engineered for cardiac repair can be retargeted to ovarian tissue perhaps using follicle-homing peptides or zona pellucida-binding ligands this convergence of evidence suggests a compelling translational pathway.
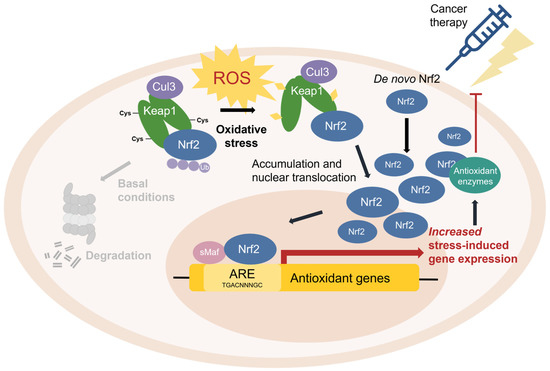
Nrf2/ARE pathway mechanism diagram [3]
Translational Potential in Regenerative Medicine
The successful demonstration of miRNA-engineered CDNs for cardiac repair has drawn increasing attention to how they could translate across systems. Gynecological disorders such as chemotherapy-induced primary ovarian insufficiency (POI) and circadian dysregulation do present one of the most compelling possibilities in this area. In much the same way as myocardial tissues do, the ovaries suffer from oxidative stress, and mitochondrial dysfunction, and also apoptosis after chemotherapeutic exposure occurs in them. For ovarian repair at specific sites, clinically approved therapies are lacking. This delivery system that is CDN-based could be adapted now for reproductive applications. That approach makes focused ovary renewal feasible. CDNs improved by miR-24-3p could target ovarian tissues precisely. This occurs when ovarian-specific peptides or follicle-homing ligands are used along with being combined with click chemistry. For women of reproductive age and women who are menopausal, such a strategy holds promise for the addressing of hormone regulation, for circadian health, and for fertility preservation.
Toward a Modular Therapeutic Platform
One overarching trend from the APSEV conference was that CDNs do envision an evolution of single-solution therapy into a customizable modular platform. Researchers presented the potential for reconfiguring CDNs through interchangeable modules tailored to address specific diseases. These modules included surface ligands intended for targeting, miRNA/protein payloads meant for function, and membrane engineering designed for circulation time in myocardial infarction, ovarian dysfunction, neurodegeneration, and so on. AI-guided drug discovery along with personalized medicine pipelines are set to integrate into this plug-and-play platform concept. CDNs also have more tissue affinity, are more flexible to manufacture, and are more immune compatible, compared to lipid nanoparticles (LNPs) or viral vectors, so systemic therapies have a more viable long-term option.
Challenges in Clinical Translation
Despite these promising developments, engineered CDNs face several important barriers prior their clinical translation. First, there are challenges to scaling up production. The current CDN isolation and purification methods are based on lab-scale ultracentrifugation, microfiltration and they have yet to be adjusted for GMP manufacture. Secondly, the immune response and long-term stability of these targeting peptides require careful attention to determine if they are safe for in vivo applications. Furthermore, the absence of standardized biodistribution and delivery efficiency metrics is a regulatory challenge and also precludes cross-study comparisons. These are issue that underline the necessity of a more effective interdisciplinary cooperation between material scientists, pharmacologists and clinician. However, as evidenced throughout the multiple presentations at APSEV 2025 (and those presented in this special issue), these hurdles do not hinder scientific progress but instead promote innovation, speed translational research and advance EV-based technologies towards realization into clinical practice.
Conclusion: A New Era at the Nano–Cellular Interface
As the frontiers of regenerative biology, nanotechnology and molecular targeting blur into each other, cell-derived nanovesicles (CDNs) are increasingly being viewed not as tools per se but rather repurposable platforms capable of bending traditional therapeutic design rules. The advances in technologies available at APSEV 2025, including miRNA-loaded vesicles and click chemistry-based targeting of Bioorthogonal chemistry are indicative for more than incremental progress. They constitute a conceptual revolution to the concept of programmable, organ-specific and patient-responsive therapies. The lesson here, whether the goal is to understand biomolecular structure or treat diseases: The future of precision medicine may not be contained in just synthetic drugs developed through reductionist principles, but might instead come from re-engineered biological messengers extracted directly from our cells. And every time such a vesicle is engineered, we are that much closer to being able to live in a world where healing can be not only targeted but smartly mobilized.
References
[1] Lv, Qingbo, et al. “Nanosponge for iron chelation and efflux: a ferroptosis‐inhibiting approach for myocardial infarction therapy.” Advanced science 11.25 (2024): 2305895.
[2] Ngo, Vy, and Martin L. Duennwald. “Nrf2 and oxidative stress: a general overview of mechanisms and implications in human disease.” Antioxidants 11.12 (2022): 2345.
Leave a comment