Dr. Chung-Liang Shih, MD
Minister of Health & Welfare
Taiwan
Today, I would like to use this space in ACTO Times to share Taiwan’s experience through a simple timeline of how our regenerative medicine policy moved from a pilot phase to a more complete, institutionalized system, and why we officially brought the Regenerative Medicine Dual Acts into force on January 1, 2026.
The “Dual Acts” refer to the Regenerative Medicine Act and the Regenerative Medicinal Products Act. For Taiwan, this is not just an update of legal text. It marks a major step forward from a technical, trial-based approach under the “Regulations of Special Medical Techniques” framework to a product-based, full life-cycle regulatory system. Looking back to 2018, when the Ministry of Health and Welfare (MOHW) announced and amended the Regulation Governing the Application of Specific Medical Examination Technique and Medical Devices (commonly known in Taiwan as the “Regulations of Special Medical Techniques”), our guiding principle has remained consistent: while responding to urgent clinical needs, we must manage risks, ensure quality, and support responsible industry development within one coherent framework. Our goal is to help innovative therapies reach patients earlier and more safely, in a way that earns public trust and, in doing so, strengthens Taiwan’s biomedical and healthcare industry.
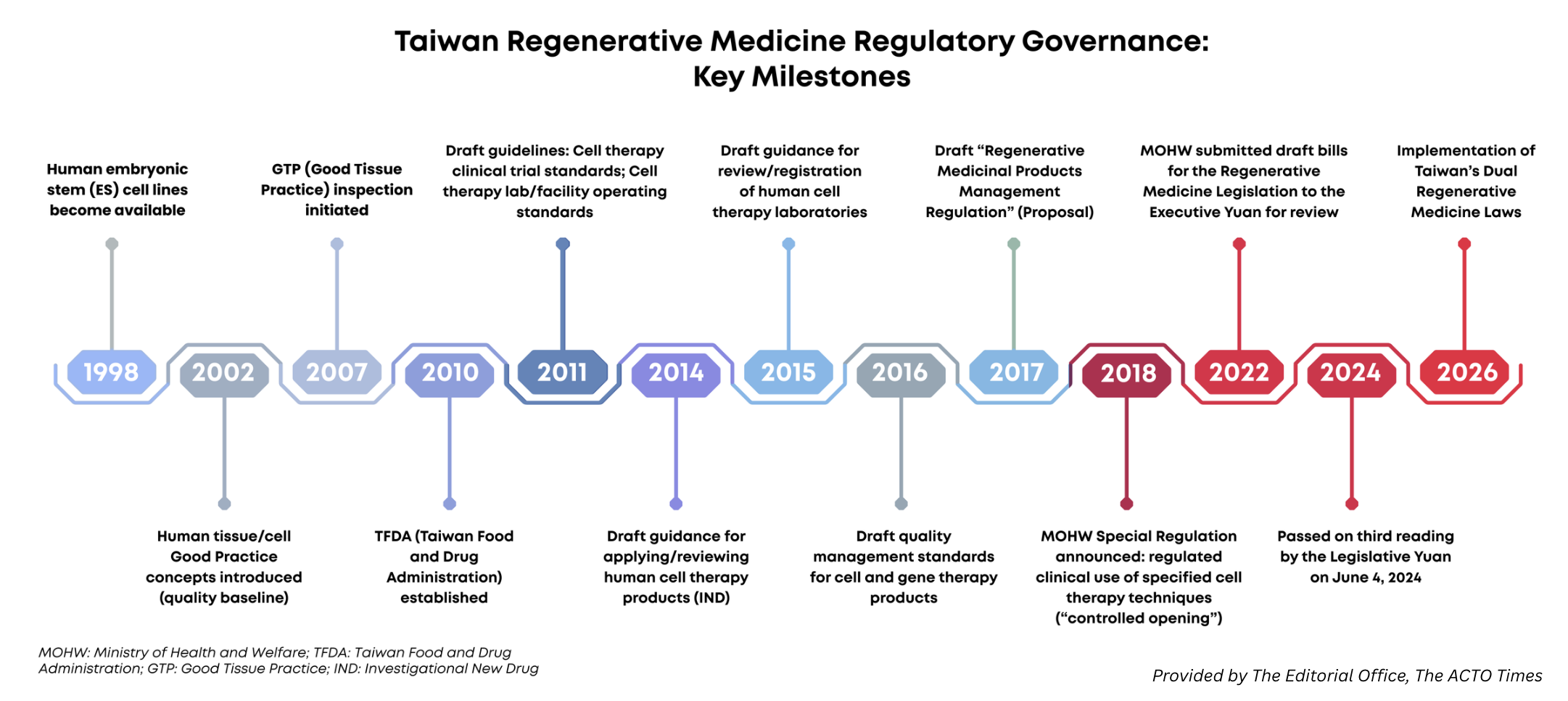
In September 2018, by amending the Regulations of Special Medical Techniques, we opened six cell therapy items for clinical use for the first time. We started with autologous (self-derived) cells for a clear reason: they are generally safer and have more predictable outcomes, based on existing evidence. At the same time, we expanded cancer-related indications from end-stage solid tumors to include patients with stage I to stage III cancers that did not respond to standard treatments, as well as certain hematologic malignancies. This enabled cell therapies to enter clinical practice within a controlled regulatory framework and to respond to patients’ urgent needs.
By the end of August 2019, when applications had reached 84 cases, we further required hospitals to adopt outcome-based, staged payment and refund mechanisms, and we announced plans to revise the regulations and build a public information portal.
In my view, progress in regenerative medicine is not only about whether we can do it, but whether we can do it responsibly. Patient protection and transparency must be built in from the start.
Two years later, in August 2020, I was invited to give a keynote address at the Taiwan Association of Cellular Therapy (TACT) Annual Meeting. Reviewing the outcomes since the Regulations of Special Medical Techniques took effect, I stated seriously that “the Regulations of Special Medical Techniques framework is reaching its limits.”
This was not a denial of its value, but a reminder: as case numbers grow and clinical use expands, a purely technical, case-by-case management approach cannot carry the system forever; we need a stronger legal structure. At that time, applications had reached 178 cases, with 46 approvals. I also made it clear that pricing should follow staged payments based on treatment course and outcomes, because risk control and patient rights must not be weakened by market enthusiasm.
In December 2020, at a press conference on cell therapy outcomes for chronic wounds at Tri-Service General Hospital, I further explained that we would announce an updated version of the Regulations of Special Medical Techniques, bring cell storage banks under management, strengthen patient registration and follow-up, and begin considering an expansion from autologous to allogeneic (donor-derived) cells. This reflected a shift in policy thinking—from “safety first” toward building a scalable supply and quality system for broader clinical use in the future.
From 2021 to 2023, we entered a critical phase: moving toward dedicated legislation and building the backbone of a long-term system.In March 2021, at the annual forum of the Formosan Association of Regenerative Medicine (FARM), I presented the key elements of the draft regenerative medicine legislation: defining regenerative medicine, regenerative products, and regenerative medical technologies; establishing an advisory committee; introducing a conditional approval mechanism; building a national-level cell bank; and allowing the Regulations of Special Medical Techniques framework to “complete its mission” and be replaced by a more comprehensive legal system.
In December 2021, we also proposed an initial concept of “three regenerative medicine laws,” emphasizing the importance of product-side regulation. If regenerative medicine is to scale up and move toward automation, the field will inevitably shift from autologous to allogeneic approaches—only then can costs come down and access expand beyond a small group of patients. Meanwhile, TFDA’s approvals of advanced therapy medical products such as Zolgensma and Kymriah also showed that Taiwan had begun building experience in reviewing advanced therapy products, laying the groundwork for a more mature regulatory system.
In May 2022, the legislative structure was refined into the “Regenerative Medicine Dual Acts” and submitted to the Executive Yuan for review. I summarized three major benefits of the proposed system: establishing a regenerative medicine fund, building a national cell bank, and using conditional approval with supporting measures to shorten development timelines and accelerate market entry.In November 2022, at a biotech forum, I again emphasized that the key regulatory focus should include manufacturing controls, sales and distribution management, and traceability of tissue and cell sources. In March 2023, responding to concerns that the Regulations of Special Medical Techniques framework might discourage formal clinical trials, I clarified that, while applications had reached 400 and about 190 had been approved, the number of INDs for regenerative medicinal products continued to rise. In other words, the system did not stop R&D; instead, it helped us find a more practical balance between clinical needs and development pathways.
In an April 2023 interview with GeneOnline, I further explained that regenerative medicine must address future trends, including allogeneic cells, gene modification, and derivatives (including exosomes). High-risk therapies require longer follow-up and stronger safety surveillance. The policy principle behind conditional approval is to meet patient needs while appropriately controlling risks and respecting patient autonomy.
From 2024 to 2026, the focus shifted to implementation and responsible commercialization. After years of effort, the Dual Acts were passed on June 4, 2024, and were set to take effect on January 1, 2026. In August 2024, at the BTC conference, I explained that the passage of the Dual Acts was a major achievement in Taiwan’s regulatory development. Their implementation will support clinical use and commercialization, and will also connect with broader infrastructure such as biomedical data governance and precision medicine.
In August 2025, at the opening of a regenerative medicine clinical trial training program, I emphasized that the Regenerative Medicine Act is not only a regulatory milestone but also signals that Taiwan’s National Health Insurance (NHI) system is beginning to seriously address reimbursement strategies and cost-effectiveness for new therapies.
At that time, six regenerative medicinal products had already been approved by TFDA, and the NHI was working through parallel review, expanded budgets, and stronger health technology assessment to help patients benefit earlier while maintaining safety and effectiveness. In late 2025, just before the Dual Acts took effect, I also highlighted another key priority: advertising control and preventing “Bad money drives out good (Gresham’s Law).
I specifically pointed to the disorder in the exosome market and warned that illegal efficacy claims would face heavy penalties—up to NT$20 million. At the same time, we ensured a seamless transition for existing cases under the Regulations on Special Medical Techniques.After the Dual Acts took effect, on January 6, 2026, I again explained in a policy forum that the core spirit of the Dual Acts is full life-cycle management: enabling earlier clinical access through conditional approval for therapies addressing high unmet medical needs, while controlling risks through institutionalized quality and safety monitoring.
In subsequent public talks and media interviews, I also emphasized a key goal for 2026: to see regenerative medicine products truly move into the stage of formal approval and market entry. TFDA will form dedicated support teams to help promising products navigate regulatory and review processes and accelerate approval.
To summarize in one sentence: the meaning of the Dual Acts is not simply “upgrading” the Regulations of Special Medical Techniques framework into higher-level laws. It is about building a complete ecosystem—from clinical pilot use, data accumulation, and risk governance, to product development, scalable manufacturing, and market oversight.
Our policy pathway can be described as follows: start with autologous cells within hospitals to establish safety and follow-up; gradually expand to allogeneic and more complex technologies; use conditional approval to shorten the waiting time for patients; ensure traceability and risk control through national cell banking and quality monitoring; and maintain market order through advertising and compliance enforcement.
The real challenges ahead are clear: whether real-world data collected after conditional approval can be analyzed with high quality; whether long-term follow-up and adverse event reporting can be fully implemented; whether regulatory capacity and industry support can grow together; and whether reimbursement decisions can remain transparent, consistent, and fair.
If these elements are institutionalized and executed well, 2026 will not only be the year the Dual Acts took effect, it will also be a true turning point for Taiwan, moving regenerative medicine from research to broader clinical access and from isolated cases to an affordable, well-regulated market.
Finally, Taiwan’s experience reflects our long-standing core values: We are family / Team Taiwan, and Taiwan can help. We are willing to share our experience in system-building, risk governance, and responsible industry development with all ACTO members, so that regenerative medicine can benefit more patients on a safer, more transparent, and more sustainable foundation.

Dr. Chung-Liang Shih, MD
Minister of Health & Welfare
Taiwan
Leave a comment